Significant news this week around a New England Journal epublication - Forde et al. - and a matching op ed by Lovly. Find them here and here. See news stories at Medscape, at Healio, at MedPage (where, "quantum leap" is the subheadline.)
In a phase III RCT, 358 patients were randomized to neo-adjuvant (pre-lobectomy) chemotherapy or chemotherapy plus nivolumab (Opdivo).
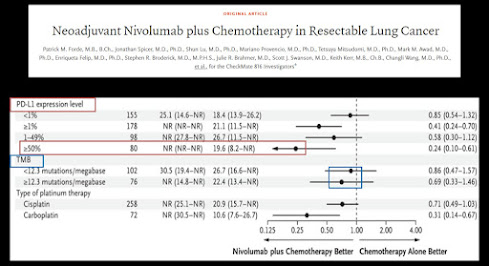
Event free survival was 32 vs 21 months, favoring the Opdivo group, and there was a strong trend toward increased survival (OR 0.6, but p > .05). Pathological complete response reached 24% in the Opdivo group but was negligible (2%) in the regular group. Note that only 75-80% of patients had definitive surgery (the point of neoadjuvant therapy is to prepare the cancer and surrounding tissue for the definitive surgery). The authors add that Opdivo + platinum therapy is a significant advantage over platinum alone. Patients who were EGFR or ALK positive were not entered into the study.
The article made minimal note of biomarkers, although both PDL1 and TMB were tested. The op ed and 3 news articles mentioned above didn't mention biomarkers (except to note the patients were stratified by PDL1 between the two arms). So - What about the biomarkers?
333 of 358 patients had PDL1; 178 of 358 had TMB (chart below). TMB impact was small. While the article said that all groups appeared to benefit, from the charts the high PDL1 group (N=80, PDL1>50%) had substantially more benefit (see chart below).
(Note that PDL1 data is shown twice, for <1% and >1%, and then again, among the >1%, for 1-49% and >50%.)
Conclusion - although it missed both the headlines and the stories, it looks like high-PDL1 patients garnered a disproportionate share of the benefit of the new combo.
 |
| Click to Enlarge |
__
See a detailed April 8, 2022, article on 360DX by Leo O'Connor on a new effort for better standardization of immunohistochemistry, the CASI (Consortium for Analytic Standardization of Immunohistochemistry.) Here.