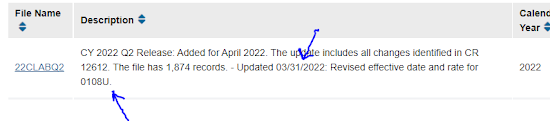
CMS has updated its ADLT test page to show a new test, 0108U, the Castle Biosciences/Cernostics "TISSUECYPHER" test for Barrett's esophagus. Castle press release here.
Find the listing here:
https://www.cms.gov/files/document/advanced-diagnostic-laboratory-tests-under-medicare-clfs.pdf
The test is priced at $2350.
Code 0108U is for "Gastroenterology, Barrett's esophagus, whole slide digital imaging, including morphometric analysis, computer assisted quantitative immunohistochemistry of 9 protein biomarkers, and morphology, FFPE tissue, algorithm as risk of progression to high grade dysplasia or cancer.”
I had not been aware of 0108U as a "covered test." (CMS prices tests with codes, whether or not they're covered.) However, it has to be a "covered test" to win ADLT status, so it must be.
Concentration of ADLTs
I tally 12 ADLT codes, with this ownership:
- 4 Castle, 2 each for Biodesix, FMI, one each for Guardant, Natera, Myriad and Veracyte. (Note that the CMS ADLT page shows a Melanoma test as pertaining to "Myriad," 0090U, but it was traded to Castle.)
Two tests are similar (FMI liquid biopsy panel 0239U, Guardant liquid biopsy panel 0242U), because they are ADLTs conferred via the FDA route to ADLT, rather than by the MAAA route which adds a "uniqueness" criterion. Those two tests are also automatically covered under NCD 90.2, which covers all FDA approved NGS CDx tests.
I initially found the conversion of 0108U to an ADLT puzzling. Creation of a new ADLT code is pretty cut and dried and follows ADLT regulations. The test has to be covered by CMS, and be a MAAA test of DNA, RNA, or protein, and be "unique" as interpreted by CMS. Then, the test is initially priced at the list price, which is carefully defined in regulation as the lowest publicly available price on the first day the test is offered to the public. OK.
Code 0108U is not so new (we're approaching 400 PLA codes), and already had a fee schedule price of $2513. I had previously thought that if a test was already priced on the CLFS, then, becoming an ADLT is still possible but it doesn't trigger a new-test pricing period. I may have misunderstood. A rule like that may have applied only prior to 1/1/2018 (see the footnote on the ADLT PDF page linked at top). The lack of a new ADLT period to an already-priced code may apply just if "payment was made on CLFS before 1/1/2018.")
Back the the MAAA definition. An ADLT of the MAAA type must be analytes of DNA, RNA, or protein (quotation from statute). CMS must have determined that the immunohistochemical measurements in the Cernostics test are a measurement of "protein" (intensity of staining measures protein).
_________
Cernostics isn't listed in the MolDx DEX registry and Castle has four tests listed, but not TissueCypher. I looked at the CMS Coverage Database for 0108U, and didn't find an entry for it. Cernostics is based in Pittsburgh, so it would bill into Novitas from that location. Cernostics was acquired by Castle in 12/2021.
Above image from: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ClinicalLabFeeSched/Clinical-Laboratory-Fee-Schedule-Files