What's Up?
There has been a boom in pharmacogenetics interest in the last several years. The Personalized Medicine Coalition (PMC) has just published a deep dive into how two catalogs of useful pharmacogenetics genes, and gene-drug pairs, compare and differ.
Concurrently, a UK report recommends wider use of PGx in the NHS health system (here).
________Background
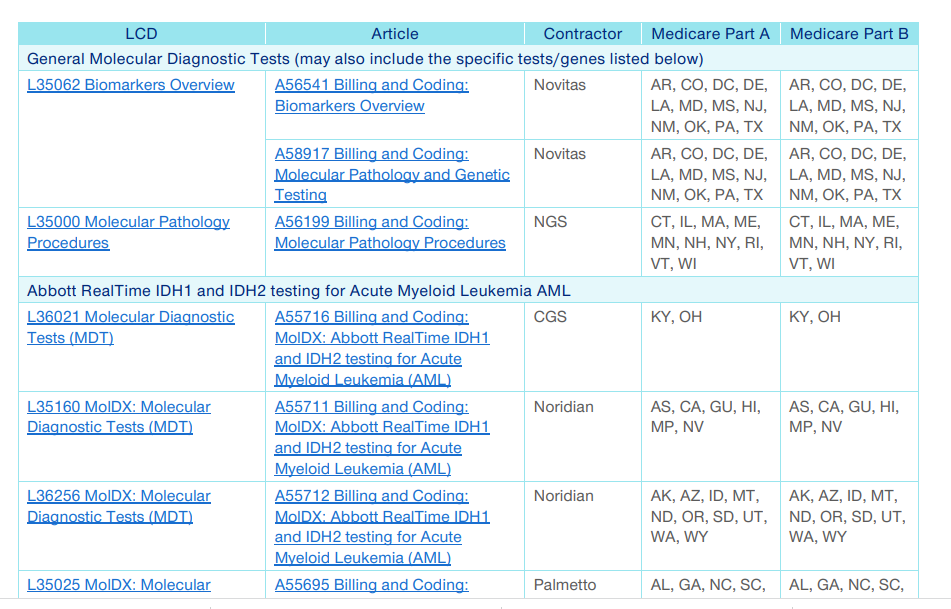
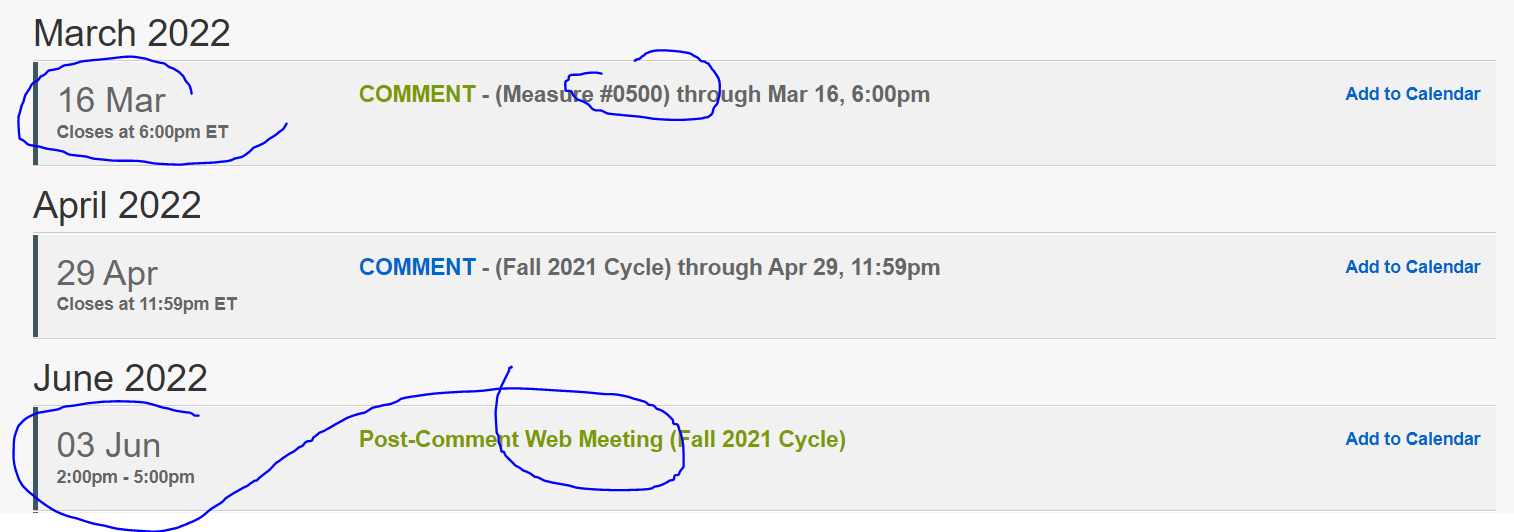
Shortly after the creation of gene-specific CPT codes in 2014, there was a huge boom in CYP gene testing in Medicare payments - the new codes generally had no controls or edits - followed by a huge fall in CYP payments as stringent LCDs appeared. (The CMS data for CYP spending around 2013-2016 looks almost like a COVID wave and plummet).
In 2020, MolDx adopted a PGx LCD which covered gene-drug pairs endorsed by either of two authorities external to Medicare, being FDA or CPIC.
My MolDx PGx blogs here, here. In late 2021, the Novitas-FCSO MACs followed with a closely similar LCD, blog here.
PMC Publishes Paper, March 2022
PMC has published a deep-dive paper comparing the two inventories references as gold standards by the LCDs, the FDA webpage for PGx genes and the CPIC website for consensus-driven PGx gene reviews.
The paper is called, Comparison of FDA Table of Pharmacogenetic Associations and Clinical Pharmacogenetics Implementation Consortium guidelines, and appears in Amer J Health System Pharm this week.
Find the new paper, open-access here.
PMC writes:
Study Finds Incongruities Between Drug-Gene Associations Listed in FDA’s Table of Pharmacogenetic Associations and Guidelines Published by Clinical Pharmacogenetics Implementation Consortium (CPIC)
Bridging Inconsistencies May Encourage More Widespread Utilization of Hallmark Personalized Medicine Tests
WASHINGTON (March 30, 2022) — A comparative analysis conducted by the Personalized Medicine Coalition and published today in the American Journal of Health-System Pharmacy reveals incongruities between the drug-gene interactions that appear in the U.S. Food and Drug Administration’s Table of Pharmacogenetic Associations and those that are referenced in widely consulted clinical guidelines published by the Clinical Pharmacogenetics Implementation Consortium (CPIC). The findings underline differing perspectives regarding the classification of certain gene-drug interactions that may deter clinicians from ordering pharmacogenetic (PGx) tests whose results would be difficult to interpret.
In comparing the drug-gene associations listed in FDA’s table with the guidelines published by CPIC, a team of authors including PMC Senior Vice President for Science Policy Daryl Pritchard, Ph.D., Atrium Health Cancer Pharmacology and Pharmacogenomics Chair Jai N. Patel, Pharm.D., PMC Program Manager Lindsay E. Stephens, and Intermountain Healthcare Executive Clinical Director of Precision Health Howard L. McLeod, Pharm.D., found that the same drug-gene associations and dosing implications were reported for only five of the 126 drugs that were listed by one or both sources. An additional 34 drugs were listed by both sources but with differing gene associations and dosing implications. The remaining 87 drugs were listed by only one of the two sources.
“This study reveals conflicting information regarding potential drug-gene interactions that undoubtedly cause confusion among health care providers, thereby slowing the integration of pharmacogenomics, one of the pillars of personalized medicine, into clinical care,” said PMC President Edward Abrahams.
British Recommendation: More PGx
See an open access article at Genomeweb about a British report 951pp) recommending more PGx in the UK.
https://www.genomeweb.com/policy-legislation/uk-report-urges-widespread-nhs-uptake-pharmacogenomics
Report online -