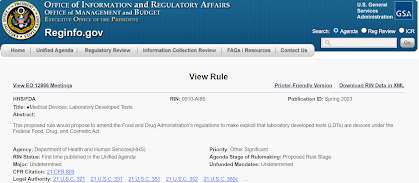
In a dry and brief federal regulations announcement, FDA has formally announced that it plans to issue regulations about LDTs, lab developed tests, very soon.
Expect proposed regulations by August 2023.
Read more about it at Reginfo.gov.
https://www.reginfo.gov/public/do/eAgendaViewRule?pubId=202304&RIN=0910-AI85
| HHS/FDA | RIN: 0910-AI85 | Publication ID: Spring 2023 |
| Title: ●Medical Devices; Laboratory Developed Tests | |
| Abstract: This proposed rule would propose to amend the Food and Drug Administration’s regulations to make explicit that laboratory developed tests (LDTs) are devices under the Federal Food, Drug, and Cosmetic Act. ## In the Trump era there were HHS memos that FDA could not regulate LDTs; see Topic #1 at a 2021 blog and see "ZIP" file there. |