On January 25, 2022, I published a blog that the ever-confusing gapfill season for lab codes is underway at CMS and its MACs. Here. By February 8, a lot more information has been posted.
The 2022 Gapfill Codes are 81349, 81560, 86408, 86409, 86413, 87426, 87811, 0018M, 0224U, 0226U, 0228U, 0245U, 0248U, 0249U, 0252U, 0253U, 0254U, 0255U, 0256U, 0257U, 0260U, 0262U, 0264U, 0265U, 0266U, 0267U, 0268U, 0269U, 0270U, 0271U, 0272U, 0273U, 0274U, 0276U, 0277U, 0278U. I count 36. MACs collect information now, submit to CMS in mid spring. CMS publishes MAC prices, and takes public comment. Having considered public comment, MACs resubmit final prices to CMS in August. The prices last for several years, until the next PAMA cycle.
NGS & Novitas/FSCO
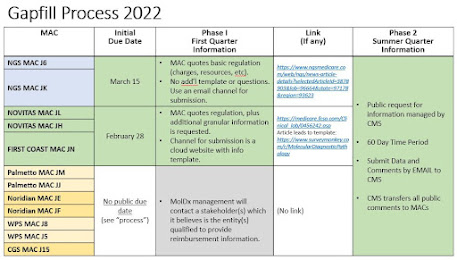
In the last few days, NGS MACs and Novitas/First Coast MACs have published articles and provide channels and due dates for submitting Gapfill lnformation.
NGS MACs ask for basic gapfill information in your own format, and submit by email by March 15.
The Novitas/FCSO MACs provide a cloud data template and offer some special detailed questions about costs, overhead costs, etc. The Novitas/FCSO MACs have a due date of February 27/28.
Gapfill in the MolDx System
The MolDx MACs (Palmetto, Noridian, WPS, CGS) operate differently.
They do not have a public article nor due date about the current national gapfill process. MolDx management will reach out proactively to a stakeholder (or plural) whom it believes appropriate to file a gapfill comment. Alone of the various MACs, MolDx does not publicize its due date on the website, which tends to make clients nervous that a due date might have been yesterday.
- If you are a stakeholder that needs to comment, and to MolDx, I would suggest reaching out to MolDx management (and by mid February) by email to see what your options are and learn what the nonpublished due date is.
- MolDx prices are critically important, since CMS sets the code price to a national "median" and the "median" is always controlled by the MOLDX block of MACs.
Summary
 |
| Click to Enlarge |
Links
Here is are the article links for First Coast and for Novitas. Each contains a link to a cloud entry form.
- https://medicare.fcso.com/Clinical_lab/0456242.asp
- https://www.novitas-solutions.com/webcenter/portal/MedicareJH/pagebyid?contentId=00246106
Here is the article link for NGS MAC:
_____
Nerd Notes
Nearly all the Category I codes under gapfill are COVID codes. 81439 is low pass NGS cytogenomics, and 81560 is a T-cell transplant test as a MAAA. I assume the COVID codes just missed the June 2020 pricing meeting, so they came to the June 2021 pricing meeting, and thus the Spring 2022 gapfill work.
Notably, the several MACs take different approaches to gathering information. A number have public articles, deadlines, and specific request methods (in the case of Novitas/FCSO). And that's what I had assumed the rule or instruction was. But in contrast, the MolDx MACs do not open any public article or website, but reach out to relevant entities for each code. As I noted above, this makes me edgy I might miss some unclear deadline and get in hot water for having missed it.
Nothing in the program manuals or regulations appear to push either way; CMS only asks MACs to price all the codes by April 1 and to use the decades-old suggested familiar information (charges, resources, etc, 42 CFR 414.508). Unlike an LCD proposal, which has comment period rules, I findno CMS statement towards the MACs in the vein of first quarter public comment (or making it easy for the public). Lacking such a command, MACs are still free to post websites, portals, deadlines, and so on, and publicize them. Certainly, I've always said and continue to say, if you're a lab test in gapfill, for gosh sakes, stay ahead of the ball and at every MAC.
It seemed weird to me (the Novitas approach seems the most natural to me) but think of Gapfill pricing like LCDs. MACs can reach out for advice to make proposed LCDs, but nothing is really required. After they do post a proposed LCD, though, there is a very well structured open public comment process.
So, CMS never refers to "public comment" now, in the initial development period for gapfill. BUT, once CMS posts the MAC proposals (hopefully in May, but sometimes weirdly late), CMS then does host a true public comment period with comment instructions, and a comment mechanism, and public due dates.
____________
I've included the text cut and pasted the NOVITAS template below the break. For the most current version, see the links just above.
###
https://www.novitas-solutions.com/webcenter/portal/MedicareJH/pagebyid?contentId=00246106
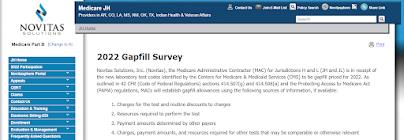
2022 Gapfill Survey
Novitas Solutions, Inc. (Novitas), the Medicare Administrative Contractor (MAC) for Jurisdictions H and L (JH and JL) is in receipt of the new laboratory test codes identified by the Centers for Medicare & Medicaid Services (CMS) to be gapfill priced for 2022. As outlined in 42 CFR (Code of Federal Regulations) sections 414.507(g) and 414.508(a) and the Protecting Access to Medicare Act (PAMA) regulations, MACs will establish gapfill allowances using the following sources of information, if available:
Novitas seeks your input on establishing pricing under the Medicare program for gapfill laboratory test codes 81349, 81560, 86408, 86409, 86413, 87426, 87811, 0018M, 0224U, 0226U, 0228U, 0245U, 0248U, 0249U, 0252U, 0253U, 0254U, 0255U, 0256U, 0257U, 0260U, 0262U, 0264U, 0265U, 0266U, 0267U, 0268U, 0269U, 0270U, 0271U, 0272U, 0273U, 0274U, 0276U, 0277U, and 0278U. To that end, Novitas kindly requests that you complete our molecular diagnostic pathology survey by March 3, 2022.
[Parts of the cloud form, clipped below]
This survey is for information gathering for the Medicare clinical laboratory fee schedule gapfill pricing process. Though additional information on analytical and clinical validity and clinical utility of a molecular pathology procedure is appreciated, benefit category and coverage determinations are a separate process. It is important to point out that some of the services billed for genetic testing may not meet the Medicare reasonable and necessary threshold for coverage.
Please note that your answers will be kept to the greatest degree of confidentiality and will not be disclosed, except as may be necessary to share with others under confidentiality agreements with CMS for the sole purpose of establishing Medicare clinical laboratory fee schedule gapfill pricing.
Your responses will be reviewed and evaluated by Novitas and if proper, set forth in its recommendations to CMS of the appropriate fees for these tests. If no response is received, the fee will be determined by evaluating the charges, payment amounts, and resources required for other tests that may be comparable or otherwise relevant.
Note: The presence of an established local payment amount does not imply coverage.
###
###
###
Cloud form, from the link in the middle of the above article. Parts of the cloud form only.
https://www.surveymonkey.com/r/MolecularDiagnosticPathology
1. Checkboxes for each possible gapfill code for 2022.
2. Clinical vignette (Text box)
3. How was the service billed previously (before this new code)? (Text box)
4. Please classify as (10 LDT, (2) FDA cleared or approved IVD, (3) "modified DFA cleared/approved."
(BQ: I think this last means you started with an FDA kit and modified it significantly enough to be a different test).
5. Total charges for the test and discounts. (Text box).
6. (Cost Detail)
Please detail the underlying costs required to provide this test. Costs can be given as per specimen OR per batch of specimens (if the latter, please provide the maximum number of specimens per batch). Categories for underlying costs are as given below:
- Personnel labor costs
- Laboratory staff (e.g., accessioning personal, technicians, variant scientists)
- Professional/signatory staff (e.g., physician)
- General (NOT test specific) laboratory costs (e.g., facilities, general equipment such as pipettes, general consumables such as gloves, quality assurance, recertifications)
- Test specific costs
- Instrument(s), including associated software and bioinformatics
- Either as amortized per test/batch OR as total original purchase price
- Consumables (e.g., instrument-specific reagents)
- Test development (including validation) costs, amortized per test/batch
7. If applicable, list charges, payment amounts, and resources for other tests that may be comparable or relevant.
8. Please provide direct indirect costs associated with the test. (BQ: I think they want a ratio, like 50/50 or 60/40).
9. Please attach if you like supporting documentation.
- Laboratory staff (e.g., accessioning personal, technicians, variant scientists)
- Professional/signatory staff (e.g., physician)
- Instrument(s), including associated software and bioinformatics
- Either as amortized per test/batch OR as total original purchase price
- Consumables (e.g., instrument-specific reagents)
- Test development (including validation) costs, amortized per test/batch