For a few years, this has been the status quo for Medicare bundling of tests based on blood or tissue samples taken in the hospital outpatient environment (both hospital outpatient medical clinics and hospital outpatient surgical centers.) The rules are at 42 CFR 414.510.
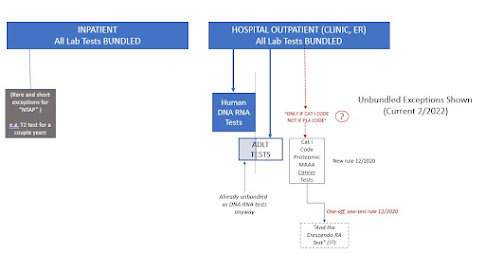
All laboratory and pathology tests are bundled to the underlying procedure or office visit, EXCEPT for MOLECULAR PATHOLOGY tests.
In practice, CMS defines molecular pathology tests as human DNA/RNA and marks these with a payable indicator in the outpatient fee schedule. Payable tests are paid by the lab that performs them (whether hospital or reference lab.)
Then, CMS adds a couple more exceptions (see 414.510). Payable are ADLT tests of type 1, which are ADLT tests because they are covered MAAA tests that have applied to be ADLT tests. Most of these are DNA/RNA tests anyway, but if it's a MAAA ADLT it doesn't have to be DNA/RNA.
And payable also are "proteomic MAAA tests for cancer." This latter was added on December 29, 2020 (see 85 Fed Reg 86251ff). My marked-up cloud copy is here. CMS opens the discussion noting it had considered this issue in 2019 policymaking and was now revisiting it in 2020.
Here's where it gets even quirkier, for two reasons.
Category I Rule.
CMS said that the proteomic MAAA tests for cancer, of which it tallied about six, had to be Category I CPT Codes (classified by AMA CPT as MAAA tests) and not PLA codes.
CMS held firm on this, despite multiple complaints. Possibly, AMA or CAP or others should complain this is weird and unfair discrimination against PLA codes. At some points, in fact, AMA has said that "once a PLA code, always a PLA code" so some tests wouldn't be eligible to graduate from PLA status to 815nn MAAA code status. (fn1)
One Non-Cancer MAAA Test Slips In!
Then, CMS did something really odd. While the regulation is for "protein based MAAA tests for cancer," CMS also included a single protein based MAAA test for rheumatoid arthritis, CPT 81490. Don't get me wrong, I'm happy this test got included, payable rather than bundled, but it's pretty darn weird it's included under a regulation written explicitly for protein based MAAA tests "for cancer" quote-unquote.
Back to the Future?
CMS says its intention in the whole Date of Service policy domain is to bundle "common or routine laboratory tests that would otherwise be packaged into OPPS payment." (p. 86254) My sense is there will increasingly be non-common and non-common advanced proteomic, functional cell culture, or other tests that will originate in the hospital specialty clinic setting.
fn1
 |
| Click to Enlarge |
