Just yesterday, I wrote about a new article in JAMA by CMS leadership on improving Medicare coverage of new technologies. Here.
Today, new news. An article by Tunis and colleagues in Health Affairs, "Improving Medicare Coverage of Innovative Technologies." Find the article here:
https://www.healthaffairs.org/content/forefront/improving-medicare-coverage-innovative-technologies
Note that the Health Affairs blog article links through to a longer, 33-page white paper online at Tufts, here.
The article opens,
After advances and setbacks, policy makers have revived efforts to improve the Medicare coverage process for new medical devices. Industry groups and others have long argued that the existing process is inefficient and unpredictable, and that a streamlined pathway for coverage decision making is needed for novel technologies that address serious and life-threatening illness. However, more rapid coverage processes, with shorter review periods and greater reliance on intermediate or surrogate endpoints, often mean more uncertainty about a technology’s risks and benefits at the time of coverage.
The enduring challenge is how to expedite the process to ensure that Medicare beneficiaries have appropriate, timely access to novel medical technologies, while providing robust and efficient mechanisms for evidence generation in the pre- and post-coverage period.
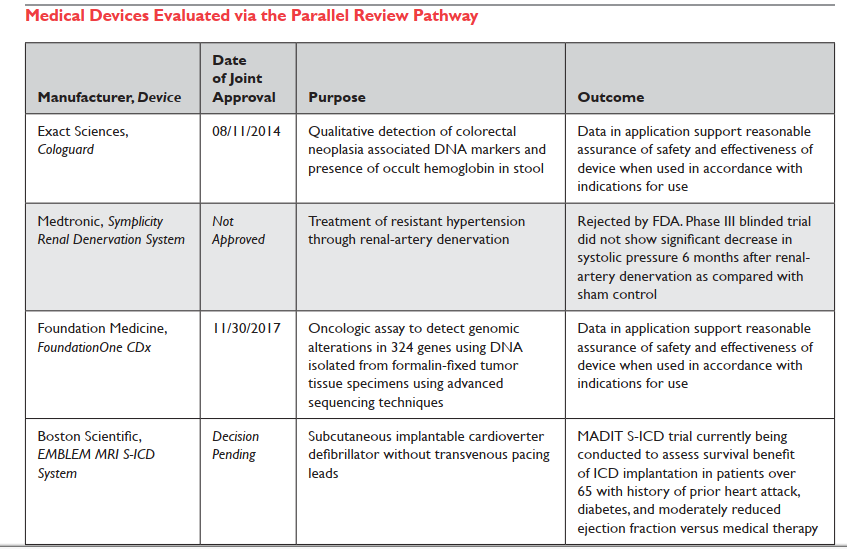 |
| Holtzman 2018 Table 2 |
In their white paper, Tunis et al. cite three possible reasons for little uptake of P.R.: (1) Company concern it will delay market entry or trigger a premature negative NCD which wouldn't occur otherwise; (2) CMS will request too many changes to the FDA plan; or (3) NCD will trigger "CED" which otherwise wouldn't come to attention. I would add a different and 4th reason: I'm aware of several companies that tried to get into the P.R. process and were turned away at the door (declined even a pilot meeting; the initial P.R. request is routed through FDA staff and got canceled there with a quick "Dear John" email reply.)
(*) If I'm reading the 2017 decision on "leadless pacemakers" correctly, it requires CED but it doesn't mention parallel review, so even if I'd read it, I wouldn't have added it to my parallel review list. I didn't see this on Google either; only in Holztman Table 2 line 4, above.
