On January 11, 2022, CMS released the proposed version of its much-debated NCD on amyloid-directed monoclonal drugs in Alzheimer's disease.
The event got widespread rapid news coverage as well as a CMS press release. In short, both ADUHELM and future in-development Alzheimer monoclonals will be covered by CMS only in the limited and controlled setting of RCTs, and, "trials supported by NIH." The latter may be an important clause, as I discuss below. Comment is open 30 days and final decision (which may vary from this proposal) is expected April 11.
CMS press release here.
CMS NCD proposal and entry to comment portal, here.
Read the book, see the movie. I've produced a brief two minute video about the decision, at YouTube, here.
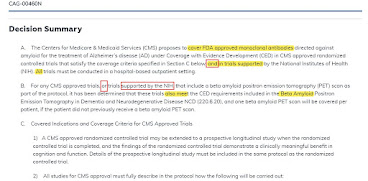
Key Points of the Proposal
Trials under Coverage with Evidence Development must be randomized controlled trials reviewed and endorsed by CMS as meeting quality criteria, or, trials supported by NIH. Trials must be hospital-based and must have populations similar to the national demographics of Alzheimer's disease (in particular, adequate minority populations.) Nothing written here limits the number or size of trials. Each patient can get one amyloid PET scan (something which is not otherwise covered by CMS.) After an RCT is run, the patients may continue on drug, with drug payment, in a long-term registry. As written, the conditions apply to future Alzheimer monoclonals in the amyloid class, even if they might have stronger primary trial outcomes than existing drugs.
At least some of the supported trials will overlap with FDA trials required by the accelerated approval.
Whether CMS can do "Coverage with Evidence Development" NCD's has sometimes been controversial; here.
"Supported by NIH" - Possibly, Potentially, Maybe Broader?
Here's one small twist, and it is ONLY hypothetical. The language covers trials reviewed and endorsed by CMS, and, trials supported by NIH. If you read the grammar carefully, it sounds like trials "supported by NIH" do not have to be entered into separate review by Medicare.
Here's a theory, and it's only a theory, based on a close reading of the grammar. There are over two dozen NIH supported Alzheimer Disease Research Centers, or ADRCs. They often conduct trials partially supported by the core NIH grant funds (for management, databases, etc) and funded additionally by other sources (pharma, foundations, donors, etc). Let's say a pharma runs a registry study at one of these centers, funding 90% of it, with part of the management funding attributed to the NIH grant. This is a study supported (in part) by NIH, but might be more of the high-quality registry type, and since it is in part NIH supported, it seems to fall into a separate track from the RCT studies discussed in the NCD.
Lots of Press
Within hours, there were countless articles about the policy proposal.
- NYT here.
- NPR here.
- USA Today here.
- AP here. Fox here.
- Axios here.
- The Hill here.
- Website "Being Patient," titled, "Why Everyone's Mad" here.
- Flurry of coverage at STAT open access - here, here. Subscription here, here, here.
- Rachel Sachs covers the decision for Health Affairs here.
- Excellent subscription article on trial design at Endpoints, here.
- For more on Biogen's response, on January 13 at Endpoints, here. BioPharmaDive here.
___
Nerd Note.
Billing, blinding. As if this is the first "nerd note" in this blog. CMS proposes randomized controlled trials, which are often blinded (doctor & patient), but how do you bill them? Coverage with Evidence Development pays for "the drug." It pays for "the treatment" under CED. Do just half the patients get a copay? How does the hospital bill for "the drug" in half the patients don't get the drug, but placebo? Does CMS pay $14,000 for all patients, knowing that 50% got a $28,000 drug? I think CMS has wrestled this bear to the ground before, but it's a gnarly one. And the doctor's office, if an infusion, can't bill for the drug unless he buys and takes ownership of the drug, which may be true for the hospital as well. And CMS isn't paying Biogen for the drug, presumably, it pays doctors and hospitals for drugs. CMS has rarely done any placebo RCTs on drugs and even rarer, or never, so there are some pitfalls here to figure out. CMS has the authority to alter payment systems used within clinical trials (with an eye to blinding etc), SSA 1833(w), but it still has to figure out HOW to do so. This provision came from the MIPPA 2008 Section 184.
CED policy. Long 2014 CMS essay about the topic, here. Short HHS Advisory Opinion in January 2021 (Trump Admin) that CED was illegal - later deleted by Biden - here. Full article by me here.
Equity. Even without considering the copay difference and "blinding," for the many minority or very poor patients, even $50 copays are too high, let alone the 20% or over $5000 copay for Aduhelm. That is another barrier to entry for CED-based equitable enrollment. STAT interviews experts on equity issues here.
Last Summer's Public Comments. CMS took public comment on the plan to do an NCD of some kind, last summer. CMS also hosted two audio sessions. The transcript of one of the audio sessions is online at CMS here. The other listening session seems to have resulted in no transcript.