Big news on February 15, 2023, that Natera receives MolDx MRD coverage for its circulating tumor DNA test "Signatera" in the indication, breast cancer.
- See the Natera press release, here.
- Open access at Genomeweb here.
- Investors Business Daily here.
- See a pending "Empowered with Meg Ryan" cancer information episode on the topic here.
- From back in December, see a San Antonio Breast Conference press release here.
According to the press release, the coverage applies to both adjuvant therapy decisions and MRD monitoring, and applies to patients with IIb or higher breast cancer stage. It applies across HR positive, or Her2 positive, or Triple Negative cases. Interestingly, the press release in particular highlights data published in 2019 as Coombes et al. But they also remark, "This study is one of several that support the use of Signatera in breast cancer, and one of over 40 peer-reviewed publications across solid tumors."
Natera presented at JP Morgan in mid January, and their year-end investor call will be February 28, 2023.
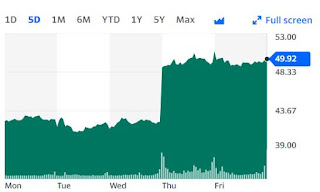
The stock shot up vertically from $42 to $49 on Thursday morning. The jump in market cap was roughly from $4.5B to $5.4B, or a billion dollars upward.
MolDx's LCD for MRD allows coverage in both solid tumors and hematopoietic cancers, and both for MRD (unexpected recurrence) monitoring and drug-therapy decisions. However, MolDx grants coverage slowly and carefully on a test-by-test basis. Most of the known coverage to date is for the Signatera bespoke tumor detection test (for colorectal cancer, for bladder cancer, and now for breast cancer.) Guardant has coverage for colorectal cancer, and Adaptive Biotechnologies for some hematopoietic cancers.
There are also negatives. In October 2022, Neogenomics announced it was dropping out of the MRD coverage race for its colorectal RaDaR product, but continuing to pursue MRD for a breast cancer project - here. We know that Neogenomics had submitted from MRD coverage in January, per another press release.